Write the resonance structures for SO3 , NO2 and NO3-
Solution and Explanation
Resonance:
Resonance occurs when a molecule or ion can be represented by two or more Lewis structures that differ only in the arrangement of electrons and not in the positions of atoms. The actual structure is a resonance hybrid of all possible structures.
(a) Resonance structures of SO3
Sulphur trioxide has three equivalent resonance structures. In each structure, one S=O double bond and two S–O single bonds are present. The double bond is delocalised among the three oxygen atoms.

(b) Resonance structures of NO2
Nitrogen dioxide has two resonance structures. The unpaired electron and the N=O double bond alternate between the two oxygen atoms.

(c) Resonance structures of NO3 minus ion
The nitrate ion has three equivalent resonance structures. In each structure, one N=O double bond and two N–O single bonds are present. The negative charge is delocalised over the three oxygen atoms.

Conclusion:
Thus, resonance structures explain the delocalisation of electrons in SO3, NO2, and NO3 minus ion, leading to greater stability of these species.
Top Questions on Kossel-Lewis Approach to Chemical Bonding
- Write Lewis symbols for the following atoms and ions: \(S\) and \(S ^{2-}\) ; \(Al\) and \(Al ^{3+}\) ; \(H\) and \(H ^-\)
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Explain the formation of a chemical bond.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
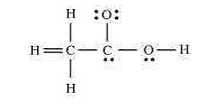
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
- K and S
- Ca and O
- Al and N.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Want to practice more? Try solving extra ecology questions todayView All Questions