Question:medium
Explain the formation of a chemical bond.
Explain the formation of a chemical bond.
Updated On: Jan 20, 2026
Show Solution
Solution and Explanation
A chemical bond is an attractive force that holds atoms or ions together in a chemical species.
Atoms combine to form bonds mainly to attain a more stable, lower-energy state, often by achieving a noble gas–like configuration of valence electrons (octet or duplet).
Octet Concept (LaTeX)
Many main-group atoms tend to adjust their valence electrons so that they have 8 electrons in their outermost shell: \[ \text{Stable configuration} \;\Rightarrow\; 8 \text{ valence electrons (octet)} \]
How Bonds Are Formed
- Ionic bond: Formed by complete transfer of electron(s) from a metal atom to a non-metal atom. The metal forms a cation and the non-metal forms an anion, both reaching stable configurations. Example: Na -> Na^{+} + e^{-} Cl + e^{-} -> Cl^{-} Na^{+} + Cl^{-} -> NaCl}
- Covalent bond: Formed by sharing of electron pairs between two atoms so that each attains an octet (or duplet for hydrogen). Example H2 molecule: H + H -> H:H
Energy Aspect
When a bond forms, the potential energy of the system decreases because the attractive forces between nuclei and electrons dominate over repulsive forces.
This decrease in energy is released as heat or light, and the bonded state is more stable than the separated atoms.
Was this answer helpful?
0
Top Questions on Kossel-Lewis Approach to Chemical Bonding
- Write Lewis symbols for the following atoms and ions: \(S\) and \(S ^{2-}\) ; \(Al\) and \(Al ^{3+}\) ; \(H\) and \(H ^-\)
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
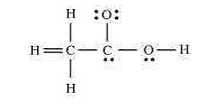
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
- K and S
- Ca and O
- Al and N.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Write the resonance structures for SO3 , NO2 and NO3-
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Want to practice more? Try solving extra ecology questions todayView All Questions