The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
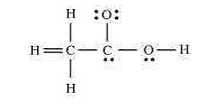
Solution and Explanation
Correct Lewis structure of acetic acid (CH3COOH):
The skeletal structure of acetic acid is correct, but the correct Lewis structure must show proper single and double bonds along with lone pairs on oxygen atoms.
Key points for the correct Lewis structure:
• The molecule contains two carbon atoms, two oxygen atoms, and four hydrogen atoms.
• The first carbon (methyl carbon) forms four single bonds with three hydrogen atoms and one carbon atom.
• The second carbon (carbonyl carbon) forms:
– a double bond with one oxygen atom (C=O)
– a single bond with the hydroxyl oxygen (C–O–H)
• Each oxygen atom has two lone pairs.
• All atoms satisfy the octet rule (hydrogen follows the duplet rule).
Correct Lewis structure representation:
H3C — C(=O) — O — H
(Each oxygen atom carries two lone pairs of electrons.)
Lewis structure diagram:

Conclusion:
Thus, the correct Lewis structure of acetic acid shows a carbonyl group (C=O), a hydroxyl group (–OH), and proper placement of lone pairs on oxygen atoms.
Top Questions on Kossel-Lewis Approach to Chemical Bonding
- Write Lewis symbols for the following atoms and ions: \(S\) and \(S ^{2-}\) ; \(Al\) and \(Al ^{3+}\) ; \(H\) and \(H ^-\)
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Explain the formation of a chemical bond.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
- K and S
- Ca and O
- Al and N.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Write the resonance structures for SO3 , NO2 and NO3-
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Want to practice more? Try solving extra ecology questions todayView All Questions