Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:- K and S
- Ca and O
- Al and N.
- K and S
- Ca and O
- Al and N.
Solution and Explanation
Lewis symbols and electron transfer:
Lewis symbols represent the valence electrons of atoms using dots around the atomic symbol. Electron transfer from a metal to a non-metal results in the formation of cations and anions.
(a) Potassium (K) and Sulphur (S)
Potassium has 1 valence electron and sulphur has 6 valence electrons. Two potassium atoms each donate one electron to sulphur.
K· + ··S·· + ·K → K+ + K+ + [··S··]2−
Thus, K+ and S2− ions are formed.
(b) Calcium (Ca) and Oxygen (O)
Calcium has 2 valence electrons and oxygen has 6 valence electrons. Calcium transfers both electrons to oxygen.
Ca·· + ··O·· → Ca2+ + [··O··]2−
Thus, Ca2+ and O2− ions are formed.
(c) Aluminium (Al) and Nitrogen (N)
Aluminium has 3 valence electrons and nitrogen has 5 valence electrons. Three electrons are transferred from aluminium to nitrogen.
Al··· + ··N··· → Al3+ + [··N··]3−
Thus, Al3+ and N3− ions are formed.
Conclusion:
Lewis symbols clearly show the transfer of valence electrons from metals to non-metals, leading to the formation of stable cations and anions.
Top Questions on Kossel-Lewis Approach to Chemical Bonding
- Write Lewis symbols for the following atoms and ions: \(S\) and \(S ^{2-}\) ; \(Al\) and \(Al ^{3+}\) ; \(H\) and \(H ^-\)
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Explain the formation of a chemical bond.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
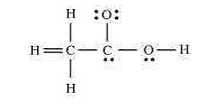
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Write the resonance structures for SO3 , NO2 and NO3-
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Want to practice more? Try solving extra ecology questions todayView All Questions