Question:medium
Which of the following has a square pyramidal shape?
Which of the following has a square pyramidal shape?
Updated On: Jan 13, 2026
PCI5
BrF5
PF5
[Ni(CN)4]2-
Show Solution
The Correct Option is B
Solution and Explanation
\(BrF_5\) (Bromine pentafluoride) exhibits a square pyramidal molecular geometry. This is attributed to the presence of five electron domains surrounding the central bromine atom, one of which is a lone pair. Consequently, Bromine pentafluoride possesses 1 lone pair and 5 bonding pairs, leading to an octahedral electron geometry and a square pyramidal molecular shape.
\(BrF_5\) has 1 lone pair and 5 bond. Therefore, geometry is octahedral, shape is square pyramidal.
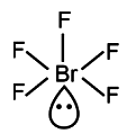
Accordingly, option (B) which states BrF5 is the correct choice.
Was this answer helpful?
0
Top Questions on Hybridisation
- The hybridisation scheme for the central atom includes a d-orbital contribution in
- BITSAT - 2024
- Chemistry
- Hybridisation
- sp\(^3\) d\(^2\) hybridisation is not displayed by:
- BITSAT - 2024
- Chemistry
- Hybridisation
Hybridisation and geometry of [Ni(CN)$_4$]$^{2-}$ are
- BITSAT - 2024
- Chemistry
- Hybridisation
- How many of the following compounds have \(sp^3\) hybridized central atoms?
\(H_2O, NH_3, SiO_2, SO_2, CO, CH_4\) and \(BF_3\)- JEE Main - 2024
- Chemistry
- Hybridisation
- Want to practice more? Try solving extra ecology questions todayView All Questions
