Question:medium
Uracil is a base present in RNA with the following structure $\%$ of $N$ in uracil is ___
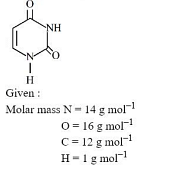
Uracil is a base present in RNA with the following structure $\%$ of $N$ in uracil is ___
Updated On: Mar 31, 2026
Show Solution
Correct Answer: 25
Solution and Explanation
To find the percentage of nitrogen (N) in uracil, we first need to calculate the molecular weight of uracil by adding up the atomic masses of all atoms present in the molecule.
Molecular formula of uracil: C4H4N2O2
1. Calculate the total molar mass:
- Molar mass of C (carbon) = 12 g/mol
- Molar mass of H (hydrogen) = 1 g/mol
- Molar mass of N (nitrogen) = 14 g/mol
- Molar mass of O (oxygen) = 16 g/mol
Total molar mass of uracil = (4 × 12) + (4 × 1) + (2 × 14) + (2 × 16)
= 48 + 4 + 28 + 32
= 112 g/mol
2. Calculate the total mass of nitrogen in uracil:
Total nitrogen mass = 2 × 14 = 28 g/mol
3. Calculate the percentage of nitrogen in uracil:
Percentage of N = (Total nitrogen mass / Total molar mass of uracil) × 100
= (28 / 112) × 100
= 25%
The percentage of nitrogen in uracil is confirmed to be within the specified range of 25 to 25.
Molecular formula of uracil: C4H4N2O2
1. Calculate the total molar mass:
- Molar mass of C (carbon) = 12 g/mol
- Molar mass of H (hydrogen) = 1 g/mol
- Molar mass of N (nitrogen) = 14 g/mol
- Molar mass of O (oxygen) = 16 g/mol
Total molar mass of uracil = (4 × 12) + (4 × 1) + (2 × 14) + (2 × 16)
= 48 + 4 + 28 + 32
= 112 g/mol
2. Calculate the total mass of nitrogen in uracil:
Total nitrogen mass = 2 × 14 = 28 g/mol
3. Calculate the percentage of nitrogen in uracil:
Percentage of N = (Total nitrogen mass / Total molar mass of uracil) × 100
= (28 / 112) × 100
= 25%
The percentage of nitrogen in uracil is confirmed to be within the specified range of 25 to 25.
Was this answer helpful?
3
Top Questions on Nucleic Acids
- Identify the polarity from ‘X’ to ‘Y’ in the mRNA segment shown. Mention how many more amino acids can be added to the polypeptide that is being translated and why.
- CBSE Class XII - 2024
- Biology
- Nucleic Acids
- Assertion (A): Uracil base is present in DNA. Reason (R): DNA undergoes self-replication.
- CBSE Class XII - 2024
- Chemistry
- Nucleic Acids
Nucleotides are joined together by:
- CBSE Class XII - 2025
- Chemistry
- Nucleic Acids
- Two nucleotides are joined together by a linkage known as:
- JEE Main - 2024
- Chemistry
- Nucleic Acids
- Want to practice more? Try solving extra ecology questions todayView All Questions
