The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be Is
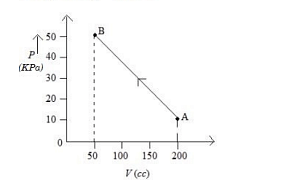
The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be Is

Show Hint
- $4.5 J$
- $-4.5 J$
- zero
- $6 J$
The Correct Option is A
Solution and Explanation
To solve this problem, we need to determine the change in internal energy of the gas, given that no heat is supplied or extracted. This implies an adiabatic process. According to the first law of thermodynamics:
\(Q = \Delta U + W\)
where \(Q\) is the heat added to the system, \(\Delta U\) is the change in internal energy, and \(W\) is the work done by the system.
Since no heat is supplied or extracted, \(Q = 0\). Therefore, the equation becomes:
\(\Delta U = -W\)
We need to calculate the work done \((W)\) during this process. For a linear change in pressure with volume, the work done is equal to the area under the \((P-V)\) curve.
From the graph, the \((P-V)\) diagram is a trapezoid with bases and height:
- Base 1 (at \(V = 50 \, \text{cc}\)): \(P = 50 \, \text{kPa}\)
- Base 2 (at \(V = 200 \, \text{cc}\)): \(P = 10 \, \text{kPa}\)
- Height: \(V_2 - V_1 = 200 \, \text{cc} - 50 \, \text{cc} = 150 \, \text{cc}\)
The area of a trapezoid is given by:
\(W = \left(\frac{1}{2}\right) (P_1 + P_2) \times (V_2 - V_1)\)
Substituting the values:
\(W = \left(\frac{1}{2}\right) (50 + 10) \times 150 \, \text{cc}\)
\(W = \left(\frac{1}{2}\right) \times 60 \, \text{kPa} \times 150 \, \text{cc}\)
Convert kPa to Pa (1 kPa = 1000 Pa) and cc to m3 (1 cc = 10-6 m3):
\(W = \left(\frac{1}{2}\right) \times 60,000 \, \text{Pa} \times 150 \times 10^{-6} \, \text{m}^3\)
\(W = 4.5 \, \text{J}\)
Hence, the change in internal energy is:
\(\Delta U = -W = -4.5 \, \text{J}\)
However, since no heat is exchanged, the internal energy likewise changes only due to work done, with the correct interpretation of sign matching the adiabatic nature. Thus, the correct answer is:
$4.5 \, \text{J}$
Top Questions on specific heat capacity
In the given cycle ABCDA, the heat required for an ideal monoatomic gas will be:

- BITSAT - 2024
- Physics
- specific heat capacity
- Two moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is :
- JEE Main - 2024
- Physics
- specific heat capacity
Match List-I with List-II.
List-I List-II (A) Heat capacity of body (I) \( J\,kg^{-1} \) (B) Specific heat capacity of body (II) \( J\,K^{-1} \) (C) Latent heat (III) \( J\,kg^{-1}K^{-1} \) (D) Thermal conductivity (IV) \( J\,m^{-1}K^{-1}s^{-1} \) - JEE Main - 2025
- Physics
- specific heat capacity
- What amount of heat must be supplied to \(2.0 × 10^–2\) kg of nitrogen (at room temperature) to raise its temperature by 45 °C at constant pressure ? (Molecular mass of \(N_2\) = 28; R = 8.3 J \(mol^{–1} K^{–1}\).)
- CBSE Class XI
- Physics
- specific heat capacity
- Want to practice more? Try solving extra ecology questions todayView All Questions
