Given below are two statements:
Statement I
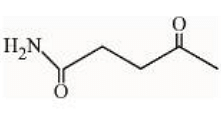
under Clemmensen reduction conditions will give

Statement II
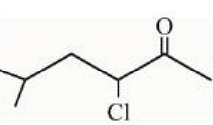
under Wolff-Kishner reduction condition will give
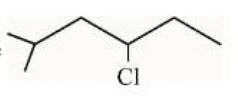
In the light of the above statements, choose the correct answer from the options given below:
Given below are two statements:
Statement I

under Clemmensen reduction conditions will give

Statement II

under Wolff-Kishner reduction condition will give

In the light of the above statements, choose the correct answer from the options given below:
Show Hint
Clemmensen Reduction: Reduces ketones or aldehydes to alkanes under acidic conditions.
Wolff-Kishner Reduction: Reduces ketones or aldehydes to alkanes under basic conditions, but reactive substituents like halogens may undergo side reactions.
- Statement I is true but Statement II is false
- Statement I is false but Statement II is true
- Both Statement I and Statement II are true
- Both Statement I and Statement II are false
The Correct Option is A
Solution and Explanation
Let's analyze both statements based on the given chemical processes:
- Statement I: The reaction of the first compound under Clemmensen reduction conditions.
The Clemmensen reduction is used to reduce ketones or aldehydes to alkanes using zinc amalgam and hydrochloric acid. In this reaction, the carbonyl group is completely reduced, removing the oxygen and saturating the carbon chain.
Under Clemmensen reduction, the given ketone will be reduced to an alkane:
This statement is true.
- Statement II: The reaction of the second compound under Wolff-Kishner reduction condition.
The Wolff-Kishner reduction is also a method used to convert carbonyl groups to alkanes, using hydrazine and strong base like KOH, with heat. However, it typically requires the compound to be a carbonyl containing group.
The given structure already appears to be an alkene. Applying Wolff-Kishner reduction to such a compound will not convert it further into an alkane like shown below:
This statement is false since it implies a reaction not applicable to the given structure.
Thus, the correct answer is: Statement I is true but Statement II is false.
Top Questions on Aldehydes, Ketones and Carboxylic Acids
The number of \(\pi\)-bonds present in benzoic acid is:
- MHT CET - 2024
- Chemistry
- Aldehydes, Ketones and Carboxylic Acids
- Which of the following reagents is used in Rosenmund reduction?
- MHT CET - 2025
- Chemistry
- Aldehydes, Ketones and Carboxylic Acids
- Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
- MHT CET - 2025
- Chemistry
- Aldehydes, Ketones and Carboxylic Acids
- Write the structures of A, B, and C in the following reactions:

- CBSE Class XII - 2024
- Chemistry
- Aldehydes, Ketones and Carboxylic Acids
- Want to practice more? Try solving extra ecology questions todayView All Questions
