Define octet rule. Write its significance and limitations.
Solution and Explanation
Octet Rule:
The octet rule states that atoms tend to combine in such a way that they attain eight electrons in their outermost shell, thereby achieving a stable noble gas configuration. This is accomplished by loss, gain, or sharing of electrons.
Significance of the Octet Rule:
1. Explains chemical bonding
The octet rule helps in understanding why atoms form chemical bonds
and how ionic and covalent bonds are formed.
2. Predicts stability of atoms
Atoms with complete octets are more stable.
This explains the low reactivity of noble gases.
3. Helps in writing Lewis structures
The rule is useful for drawing Lewis dot structures of molecules
and predicting valency of elements.
4. Basis for valence concept
The octet rule provides a simple explanation for valency
in many main group elements.
Limitations of the Octet Rule:
1. Incomplete octet
Some atoms do not complete their octet.
Examples include:
BeCl2 (Be has only 4 electrons),
BF3 (B has only 6 electrons).
2. Expanded octet
Elements of the third period and beyond can have more than eight electrons.
Examples include:
PCl5, SF6.
3. Odd-electron molecules
Molecules with an odd number of electrons do not obey the octet rule.
Examples include:
NO, NO2.
4. Does not explain shape or bond strength
The octet rule cannot predict molecular geometry,
bond lengths, or bond energies.
Conclusion:
The octet rule is a simple and useful guideline for understanding chemical bonding, but it has several exceptions and limitations. Hence, it cannot be applied universally to all molecules.
Top Questions on Kossel-Lewis Approach to Chemical Bonding
- Write Lewis symbols for the following atoms and ions: \(S\) and \(S ^{2-}\) ; \(Al\) and \(Al ^{3+}\) ; \(H\) and \(H ^-\)
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Explain the formation of a chemical bond.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
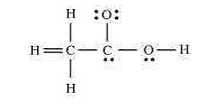
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
- K and S
- Ca and O
- Al and N.
- CBSE Class XI
- Chemistry
- Kossel-Lewis Approach to Chemical Bonding
- Want to practice more? Try solving extra ecology questions todayView All Questions