Question:medium
Based on the given figure, the number of correct statement/s is/are _______
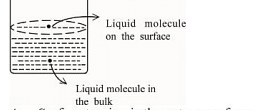
A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system
Based on the given figure, the number of correct statement/s is/are _______

A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system

A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system
Updated On: Mar 31, 2026
Show Solution
Correct Answer: 2
Solution and Explanation
To determine the number of correct statements, let's analyze each option based on surface tension concepts:
- A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk.
This is incorrect. In the bulk, a molecule experiences equal forces from all directions, cancelling out, and thus does not contribute to surface tension. - B. Surface tension is due to uneven forces acting on the molecules present on the surface.
This is correct. Molecules on the surface experience uneven forces because they are pulled more towards the bulk liquid than towards the air, creating surface tension. - C. The molecule in the bulk can never come to the liquid surface.
This is incorrect. Molecules in the bulk can move to the surface due to thermal motion and diffusion. - D. The molecules on the surface are responsible for vapour pressure if the system is a closed system.
This is correct. Surface molecules can escape the liquid to form vapor, contributing to vapor pressure in a closed system.
Correct statements: B and D, which is 2. This matches the expected range (2,2).
Was this answer helpful?
0
Top Questions on States of matter
- What is the oxidation state of Fe in \( \mathrm{Fe_3O_4} \)?
- MHT CET - 2024
- Physics
- States of matter
- Convert 72 degrees Celsius to Fahrenheit.
- MHT CET - 2024
- Physics
- States of matter
- How many lattice points are there in a Body-Centered Cubic (BCC) structure?
- MHT CET - 2024
- Chemistry
- States of matter
- What is the molar mass of a gas, if 2.5 g of the gas occupies 1.12 L at STP?
- MHT CET - 2025
- Chemistry
- States of matter
- Want to practice more? Try solving extra ecology questions todayView All Questions
