Question:medium
Which of the following will be oxidized by \(HIO_4\)?
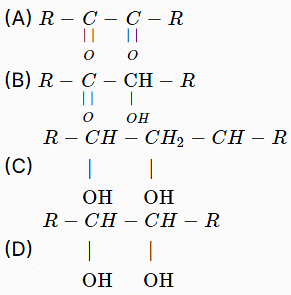
Which of the following will be oxidized by \(HIO_4\)?
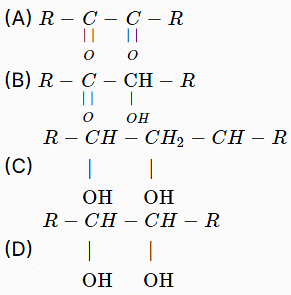
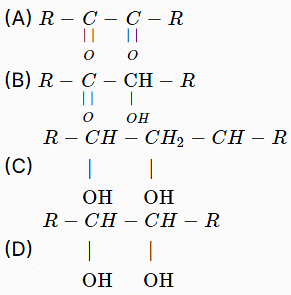
Updated On: Mar 30, 2026
- 1, 2 and 3
- 1, 3 and 4
- 1, 2 and 4
- 2, 3 and 4
Show Solution
The Correct Option is C
Solution and Explanation
The question involves identifying which compounds can be oxidized by periodic acid (\(HIO_4\)). Periodic acid is known to cleave vicinal diols and α-dicarbonyl compounds.
Let's analyze each option:
- Compound (A) is a dicarbonyl compound (\(R-C(=O)-C(=O)-R\)). \(HIO_4\) can oxidize this compound by cleaving the bond between the two carbonyl groups.
- Compound (B) is a molecule with a vicinal diol (\(R-CH(OH)-CH(OH)-R\)). Periodic acid can oxidize this compound by cleaving between the two hydroxyl groups, forming two carbonyl groups.
- Compound (C) is a non-vicinal diol (\(R-CH(OH)-CH_2-CH(OH)-R\)). \(HIO_4\) does not oxidize non-vicinal diols, so this compound will not be affected.
- Compound (D) is another vicinal diol (\(R-CH(OH)-CH(OH)-R\)) without any intervening carbon. It can be cleaved by periodic acid similar to compound (B).
Based on this analysis, compounds 1, 2, and 4 are oxidized by \(HIO_4\). Therefore, the correct answer is Option: 1, 2 and 4.
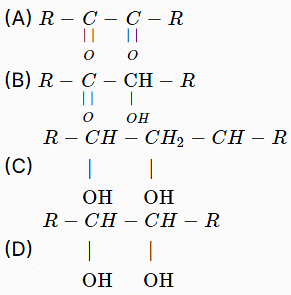
Was this answer helpful?
0
Top Questions on Oxidation Number
- What is the maximum oxidation state of an element in the periodic table?
- MHT CET - 2024
- Chemistry
- Oxidation Number
- The oxidation number of oxygen in peroxides is __.
- MHT CET - 2025
- Chemistry
- Oxidation Number
- What is the oxidation number of sulfur in sulfuric acid (H$_2$SO$_4$)?
- MHT CET - 2025
- Chemistry
- Oxidation Number
- What is the oxidation state of sulfur in \( \text{H}_2\text{SO}_4 \)?
- MHT CET - 2025
- Chemistry
- Oxidation Number
- Want to practice more? Try solving extra ecology questions todayView All Questions
