A radioactive nuclei X decays simultaneously to two nuclei Y and Z as:
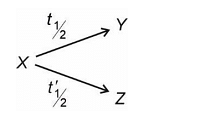
t½ is 12 minutes while t'½ is 3 minutes. Find the time in which nuclei X decays 50%.

t½ is 12 minutes while t'½ is 3 minutes. Find the time in which nuclei X decays 50%.
4.8 minutes
15 minutes
2.4 minutes
9 minutes
The Correct Option is C
Solution and Explanation
To solve the given problem, we need to determine the effective half-life of the radioactive nuclei X that decays simultaneously into two products, Y and Z.
The half-life for decay into Y (t_{1/2}) is 12 minutes and for decay into Z (t'_{1/2}) is 3 minutes. The formula for effective half-life (t_{\text{eff}}) in such cases is given by:
\frac{1}{t_{\text{eff}}} = \frac{1}{t_{1/2}} + \frac{1}{t'_{1/2}}
Substitute the given values:
\frac{1}{t_{\text{eff}}} = \frac{1}{12} + \frac{1}{3}
\frac{1}{t_{\text{eff}}} = \frac{1}{12} + \frac{4}{12} = \frac{5}{12}
Therefore:
t_{\text{eff}} = \frac{12}{5} = 2.4 \text{ minutes}
The time taken for X to decay 50% is the effective half-life, which is 2.4 minutes.
Thus, the correct answer is 2.4 minutes.

Top Questions on Radioactivity
- The half-life of a radioactive substance is 4 hours. If initially there are 256 grams, how much remains after 10 hours?
- BITSAT - 2025
- Physics
- Radioactivity
- The half-life of radioactive isotope Zn$^{65}$ is 245 days. Find the time after which activity of Zn sample remains 75% of its initial value.
- JEE Main - 2026
- Chemistry
- Radioactivity
- A radioactive sample disintegrates via two independent decay processes having half lives T₁/₂\^{(1)} and T₁/₂\^{(2)} respectively. The effective half-life, T₁/₂ of the nuclei is :
- JEE Main - 2021
- Physics
- Radioactivity
- A radio active material is reduced to 1/8 of its original amount in 3 days.
If 8×10-3 kg of the material is left after 5 days the initial amount of the material is- JEE Main - 2023
- Physics
- Radioactivity
- Want to practice more? Try solving extra ecology questions todayView All Questions
