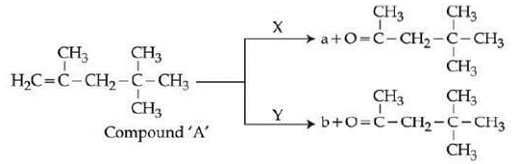
A compound A on reaction with X and Y produces the same major product but different by product a and b Oxidation of a gives a substance produced by ants X and Y respectively are :

- $KMnO _4 / H ^{+}$and dil. $KMnO _4, 273 \,K$
- $KMnO _4$, (dilute), $273 K$ and $KMnO _4 / H ^{+}$
- $KMnO _4 / H ^{+}$and $O _3, H _2 O / Zn$
- $O _3, H _2 O / Zn$ and $KMnO _4 / H ^{+}$
The Correct Option is D
Solution and Explanation
The problem involves identifying the reagents X and Y for a reaction that produces the same major product with different by-products, followed by the oxidation of one by-product to formic acid, a compound found in ants. Let's examine the options to find the correct answer.
Step 1: Understanding the Reaction
The compound A given is an alkene capable of undergoing oxidative cleavage. The major product formed from the reaction of compound A with X and Y is the same, but the by-products 'a' and 'b' are different. We also know that oxidation of one of the by-products produces a compound found in ants (formic acid).
Step 2: Evaluating the Reagent Options
- \(KMnO_4 / H^{+}\) and dilute \(KMnO_4, 273 \,K\) – Generally used for oxidation of alkenes but not suitable for producing different by-products with the characteristics needed.
- dilute \(KMnO_4, 273 \,K\) and \(KMnO_4 / H^{+}\) – Same reagents as above with roles reversed. Not very promising for producing formic acid.
- \(KMnO_4 / H^{+}\) and \(O_3, H_2O / Zn\) – Ozone followed by reduction (ozonolysis) would produce different by-products (aldehydes, ketones) which upon further oxidation with \(KMnO_4\) can produce acids such as formic acid.
- \(O_3, H_2O / Zn\) and \(KMnO_4 / H^{+}\) – Similar but reversed role. Ozonolysis can produce aldehydes/ketones, and further oxidation can lead to formic acid.
Step 3: Correct Option
The correct answer is \(O_3, H_2O / Zn\) and \(KMnO_4 / H^{+}\) because ozonolysis of alkenes can produce aldehydes or ketones, and further oxidation of aldehydes with \(KMnO_4\) can yield formic acid.
Top Questions on Haloalkanes and Haloarenes
- What is the preliminary test for nanoparticles?
- MHT CET - 2024
- Chemistry
- Haloalkanes and Haloarenes
What is the IUPAC name of the given haloarene?

- MHT CET - 2024
- Chemistry
- Haloalkanes and Haloarenes
- Which compound gives white precipitate with AgNO$_3$ in ethanol?
- MHT CET - 2025
- Chemistry
- Haloalkanes and Haloarenes
- Which of the following is most reactive towards nucleophilic substitution?
- MHT CET - 2025
- Chemistry
- Haloalkanes and Haloarenes
- Want to practice more? Try solving extra ecology questions todayView All Questions
