Which is following compound is easily attacked by electrophile?
The Correct Option is D
Solution and Explanation
To determine which compound is easily attacked by an electrophile, we need to analyze the electronic environment of each compound. Electrophiles are electron-deficient species, and they are attracted to areas of high electron density.
An electrophile will preferentially attack a compound that has electron-donating groups or conditions that increase electron density, thereby enhancing its nucleophilicity. Let's consider the compounds provided in the options:
- The first option contains an image:
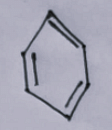
- The second option contains an image:
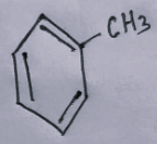
- The third option contains an image:
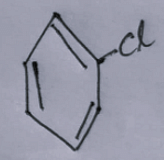
- The fourth option contains an image:
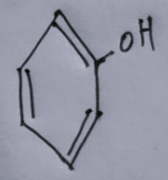
The correct answer is the fourth compound. Let's analyze why:
In the fourth compound, the presence of electron-donating groups such as alkyl groups or resonant structures that can delocalize electrons enhances the overall electron density of specific parts of the molecule, making them more susceptible to electrophilic attack.
The other compounds either have electron-withdrawing groups, such as nitro groups, which decrease electron density or are stabilized by resonance structures which do not favor electrophilic attacks as directly. Thus, they are less reactive towards electrophiles compared to the fourth option.
In summary, the correct answer is:

Top Questions on Organic Chemistry
- Which of the following compound is not allylic chloride?
- GUJCET - 2026
- Chemistry
- Organic Chemistry
- Which catalyst is used in Wacker process?
- GUJCET - 2026
- Chemistry
- Organic Chemistry
- Which product is formed on carbylamine reaction with 4-methylaniline?

- GUJCET - 2026
- Chemistry
- Organic Chemistry
- What is the correct order of basic strength in substituted amine in aqueous solution?
- GUJCET - 2026
- Chemistry
- Organic Chemistry
- Want to practice more? Try solving extra ecology questions todayView All Questions


